Understanding Cell Factor
The Product, Not the Factory
Cell Factor delivers the “end product” of regeneration—not the factory that makes it. Unlike live-cell therapies that introduce biological complexity, variability, and immunogenic risk, Cell Factor is a clinically engineered, DNA-free formulation that provides isolated signaling factors with standardized potency across every batch.
Traditional stem cell therapies are “the factory”—living cells with donor-dependent composition, regulatory classification as drugs or biologics, and inherent cellular variability. Cell Factor is “the product”—a non-cellular, shelf-stable foundation that resets cellular receptors and restores signaling precision without the complications of live biology.
This approach delivers over 400 growth factors and extracellular vesicles (EVs) that optimize cellular communication pathways. By clearing inflammatory noise and re-establishing receptor sensitivity, Cell Factor creates an environment where your body’s repair mechanisms—and any additional therapies—can function at peak efficiency.
Published research in Biomolecules (2024) confirms that acellular therapies provide “comparable regenerative potential” to stem cell-based therapies while avoiding limitations like tumor formation risk, immune rejection, and complex regulatory hurdles. Cell Factor holds FDA Section 361 tissue classification—not classified as a drug—with minimal immunogenic profile.
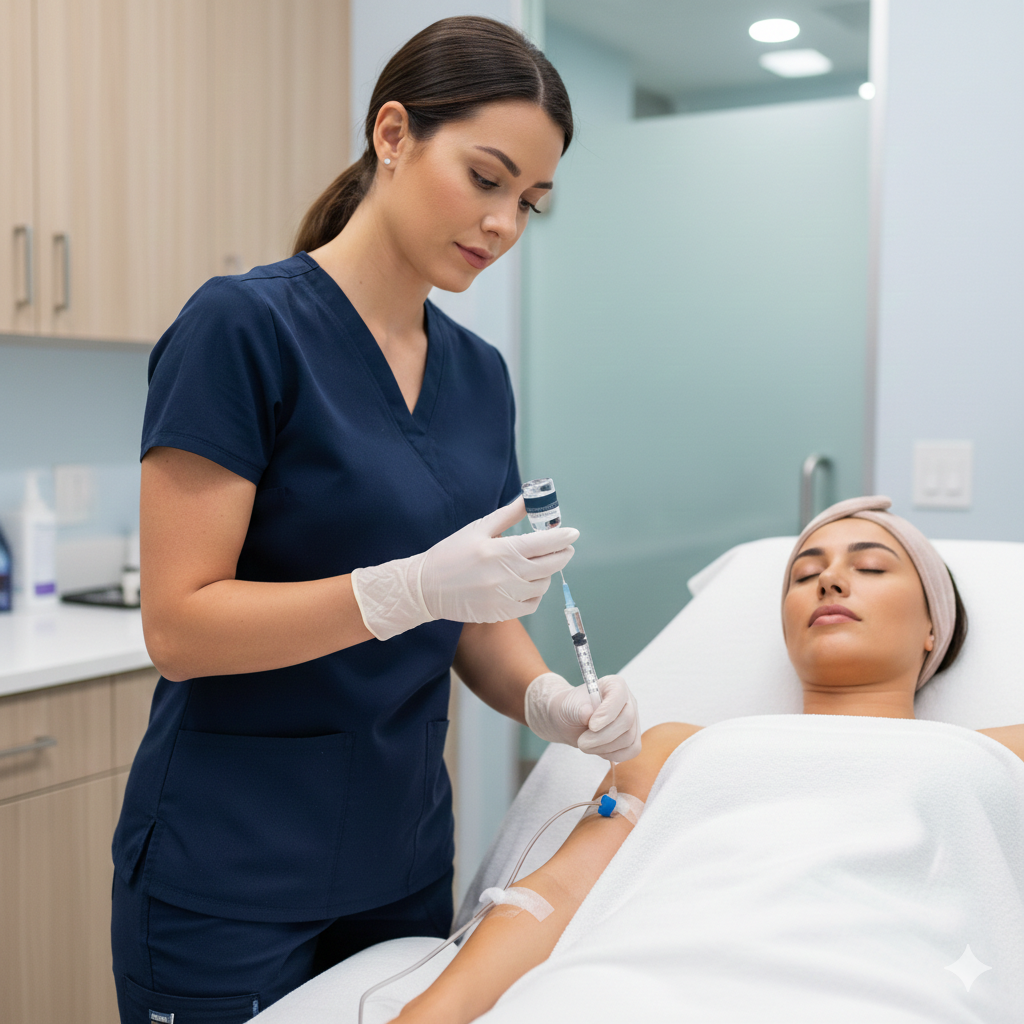
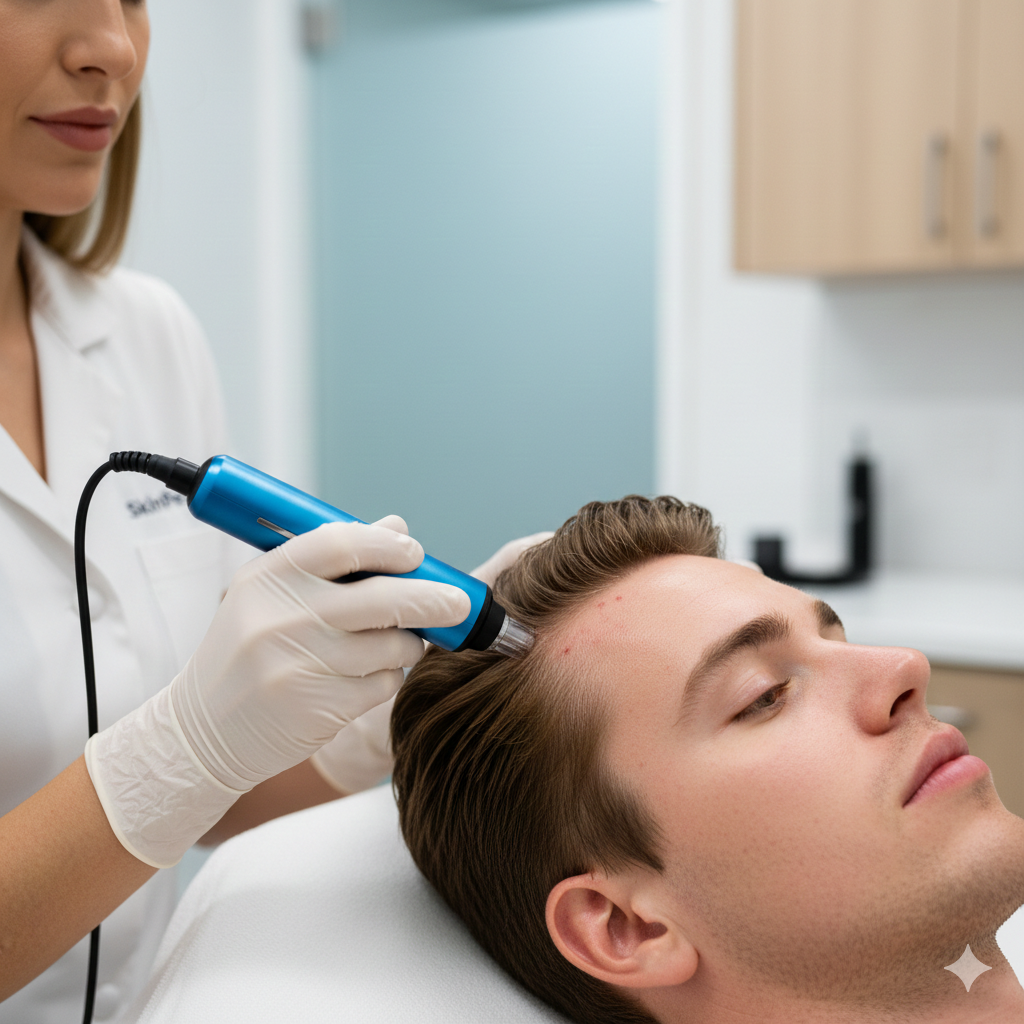
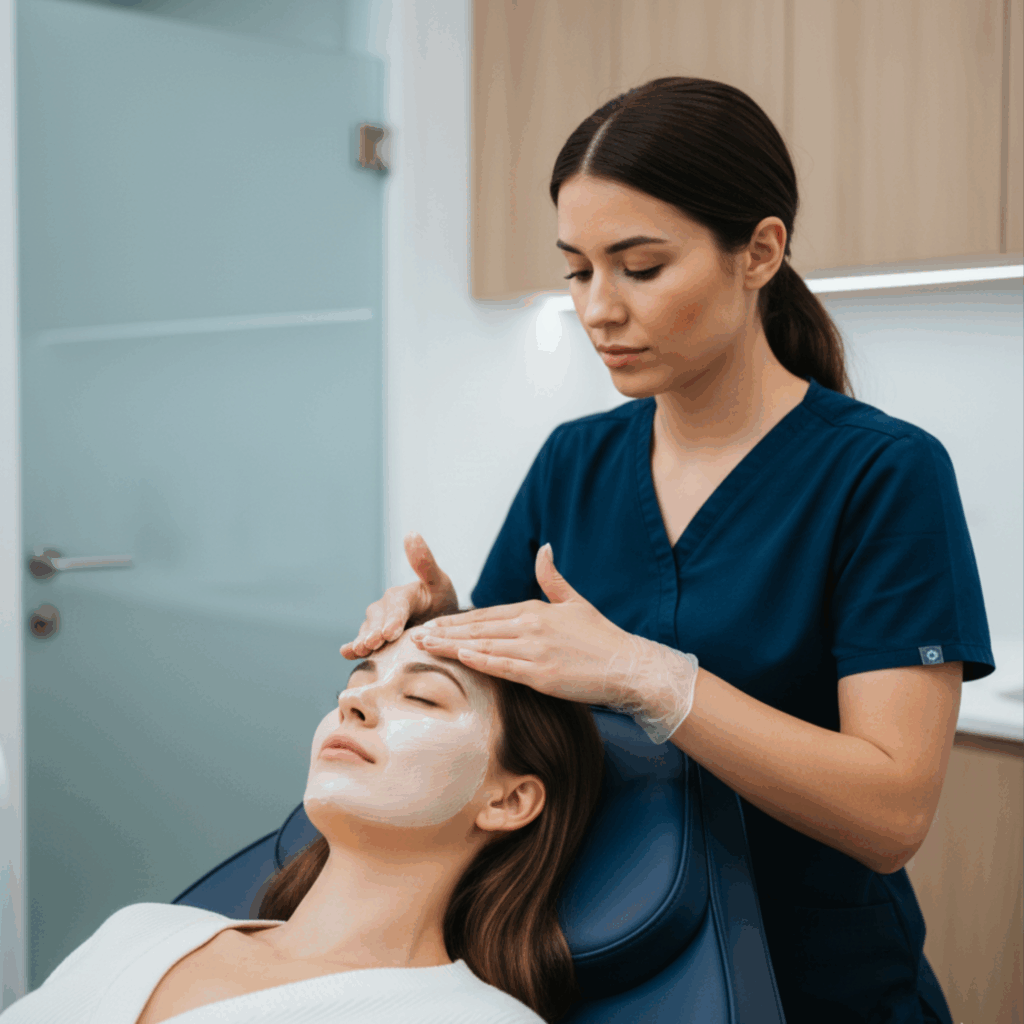
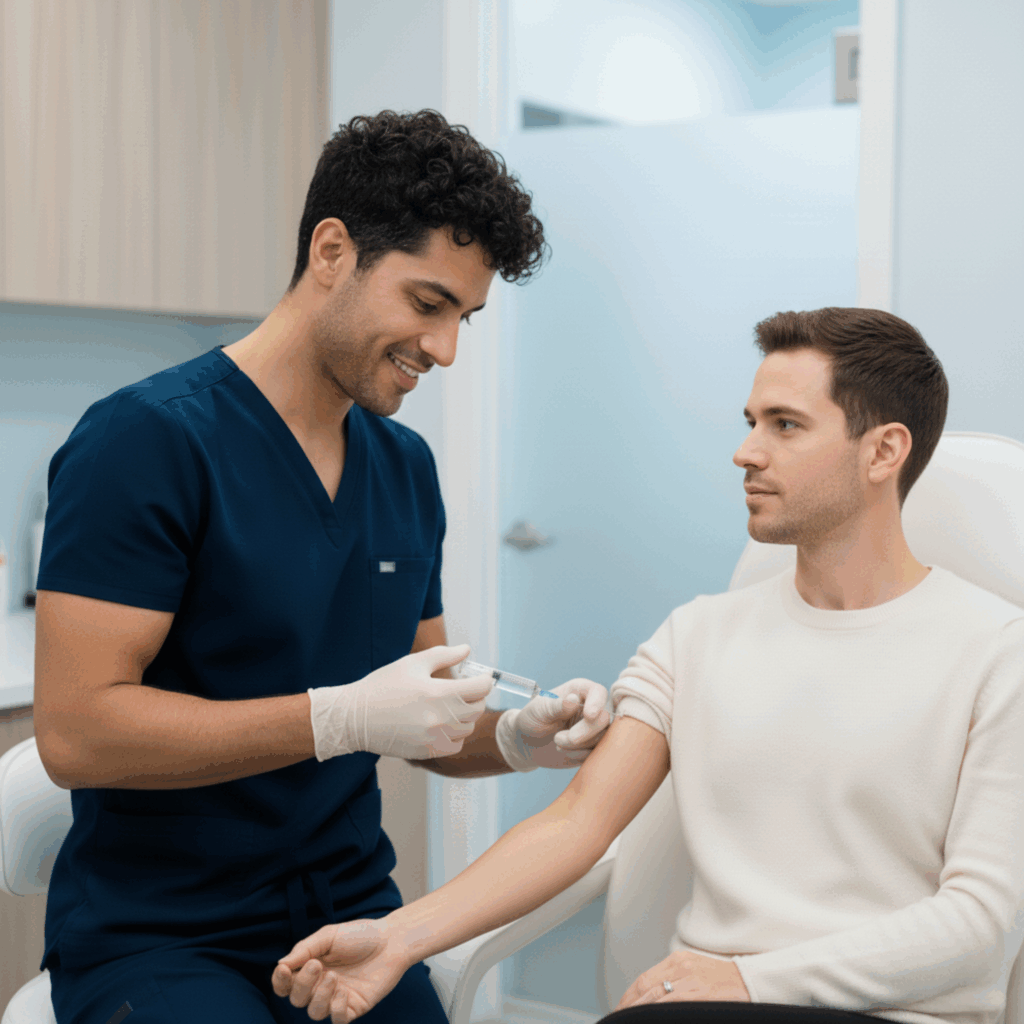
At BHRC, we integrate Cell Factor across our aesthetic and wellness treatments to amplify results and accelerate healing. Whether combined with microneedling, laser therapy, IV nutrients, or administered as standalone therapy, Cell Factor represents the next evolution in regenerative aesthetics.